THE SNAIL’S LOVE DART
When we talk about competition for mates, we often think of behaviors involving tense fights or flamboyant displays of traits that signal quality. However, sometimes the competition continues even after mating, as the sperm must outcompete sperm from other mates.
In land snails of the families Helicidae and Ariophantidae, this post-copulatory competition involves the injection of a calcium-based “love dart”. These snails are simultaneous hermaphrodites, so during mating, both partners can act as male and female, leading to …
When we talk about competition for mates, we often think of behaviors involving tense fights or flamboyant displays of traits that signal quality. However, sometimes the competition continues even after mating, as the sperm must outcompete sperm from other mates.
In land snails of the families Helicidae and Ariophantidae, this post-copulatory competition involves the injection of a calcium-based “love dart”. These snails are simultaneous hermaphrodites, so during mating, both partners can act as male and female, leading to competition over paternity. Shooting the dart forms part of the courtship ritual, in hopes of increasing the shooter’s success at paternity. The love dart is coated with sticky mucus from a gland located near the dart sac.
Using an integrative approach, Stewart and colleagues (2016) showed that the mucus of the garden snail Cornu aspersum contains a bioactive substance, dubbed the “love dart allohormone” (LDA). When the LDA is transferred to the mating partner, it induces physiological changes in the female reproductive system that reduce digestion of sperm. This socially transferred material increases the chances that the shooter’s sperm will be used for fertilization, thereby enhancing paternity success.
The study also identified numerous additional peptides in the dart mucus that may influence the mating behavior of recipient snails. Further research is needed to disentangle the functions of these peptides, as well as other LDA precursor molecules. Future work on this topic will advance our understanding of the evolution of accessory glands and their products as well as alternative reproductive strategies.
Source:
Stewart M, Wang T, Koene J, Storey KB & Cummins SF. 2016. A “Love” Dart Allohormone Identified in the Mucous Glands of Hermaphroditic Land Snails. Journal of Biological Chemistry 291:7938-7950. https://doi.org/10.1074/jbc.M115.704395
WHEN ANTS GET CLOSE…WHAT DO THEY EXCHANGE?
Ants are social insects, well known for their division of labor and caste systems, as well as their hardworking nature. They have also inspired popular culture, including sayings (“ants in your pants”), music (see “Ants marching” and “El hormiguero”), and jewelry. However, there is one curious behavior that is largely unknown to the general public (and even to many scientists) called trophallaxis, in which ants ingest liquid shared by another individual! This can occur in two different ways: as a regurgitate coming from the mouth, called stomodeal trophallaxis, or as an excretion from the rectal end of the abdomen, called proctodeal trophallaxis. Trophallaxis also occurs in earwigs, burying beetles, birds, and mammals, but we will focus on ants, an extensively studied group.
In a comprehensive review, Meurville & LeBoeuf (2021) describe how within ant colonies, trophallactic behavior occurs in multiple directions …
Ants are social insects, well known for their division of labor and caste systems, as well as their hardworking nature. They have also inspired popular culture, including sayings (“ants in your pants”), music (see “Ants marching” and “El hormiguero”), and jewelry. However, there is one curious behavior that is largely unknown to the general public (and even to many scientists) called trophallaxis, in which ants ingest liquid shared by another individual! This can occur in two different ways: as a regurgitate coming from the mouth, called stomodeal trophallaxis, or as an excretion from the rectal end of the abdomen, called proctodeal trophallaxis. Trophallaxis also occurs in earwigs, burying beetles, birds, and mammals, but we will focus on ants, an extensively studied group.
In a comprehensive review, Meurville & LeBoeuf (2021) describe how within ant colonies, trophallactic behavior occurs in multiple directions, including adult-to-adult, adult-to-larva, and larva-to-adult interactions. It even can occur with other organisms such as aphids, butterflies, and plants! The fluid that they socially exchange is a cocktail of microorganisms, proteins, nucleic acids, and small molecules. Proctodeal fluid may also include excretions from the Dufour’s gland and poison gland, as well as proteins from venom. Trophallaxis happens in different contexts such as during nourishment, decision-making, defense against pathogens, social maintenance, aggression (both within and between species), and the inoculation and maintenance of the gut microbiota.
Using an integrative approach that combined evolutionary and phylogenetic analyses with deep-learning tools, Meurville and colleagues (2025) inferred that trophallaxis evolved in two main events: first 130 million years ago (mya) in non-doryline formicoid ants and later, around 90 mya, in some ponerine ants. They identified a high number of gains and losses across the phylogeny, suggesting that social regurgitation is a highly plastic behavior. Its evolution was enabled by both the reduction of social conflict and the opportunistic inclusion of nectar and honeydew in the ant diet, which coincided with shifts toward flowering plants in terrestrial ecosystems.
Studying the macroevolutionary history of trophallaxis and its consequences is challenging, given that these types of behavior are not easily inferred from fossil evidence and are difficult to assess and quantify even in extant species. Nevertheless, further research is needed to determine the role of this social transfer in shaping the ecological success of one of the most dominant groups in the animal kingdom.
Meurville and LeBoeuf run an open-science database, “Trophallaxis behavior in ants”, which compiles known trophallactic behaviors across ant species. Please do contact them if you can contribute new observations!
Sources:
Meurville MP, Silvestro D & LeBoeuf AC. 2025.Ecological change and conflict reduction led to a social circulatory system in ants. Communications Biology 8:246. https://doi.org/10.1038/s42003-025-07688-7
Meurville MP & LeBoeuf AC. 2021. Trophallaxis: the functions and evolution of social fluid exchange in ant colonies (Hymenoptera: Formicidae). Myrmecological News 31: 1-30. https://doi.org/10.25849/myrmecol.news_031:001
WHEN YOUR VERY FIRST MEAL IS YOUR SIBLING
Nature never stops amazing us with the incredible diversity of parental strategies that have evolved to increase offspring survival. During development, mothers provide nutrients to their young in different ways, usually through a placenta, maternal tissues, or yolk stored in the egg. But, can you believe that there is an extreme case in which the food that mothers provide is the siblings that are sharing the same maternal space? This phenomenon, known as adelphophagy, is relatively rare in the animal kingdom, but has been documented in vertebrates such as sharks, as well as in invertebrates like echinoderms, insects, molluscs, and annelids.
Maternal feeding of siblings also occurs in two types of invertebrates that are unfamiliar to many of us: flatworms and ribbon worms. Both are…
Nature never stops amazing us with the incredible diversity of parental strategies that have evolved to increase offspring survival. During development, mothers provide nutrients to their young in different ways, usually through a placenta, maternal tissues, or yolk stored in the egg. But, can you believe that there is an extreme case in which the food that mothers provide is the siblings that are sharing the same maternal space? This phenomenon, known as adelphophagy, is relatively rare in the animal kingdom, but has been documented in vertebrates such as sharks, as well as in invertebrates like echinoderms, insects, molluscs, and annelids.
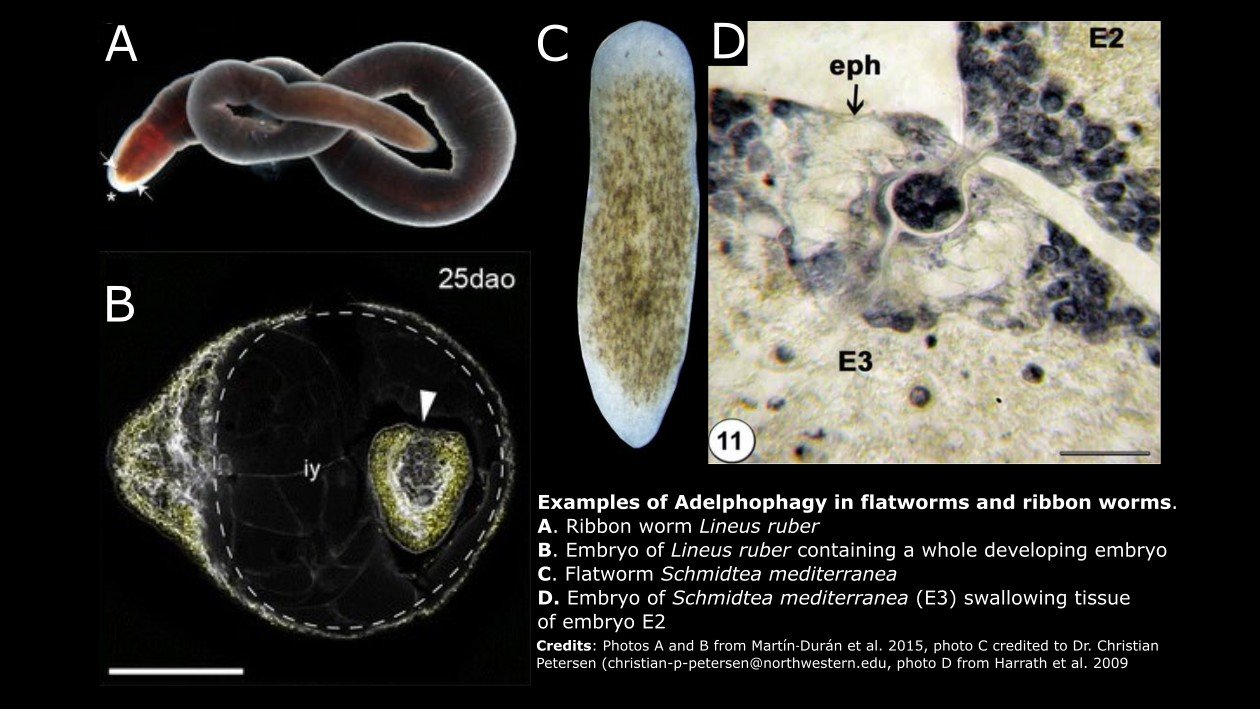
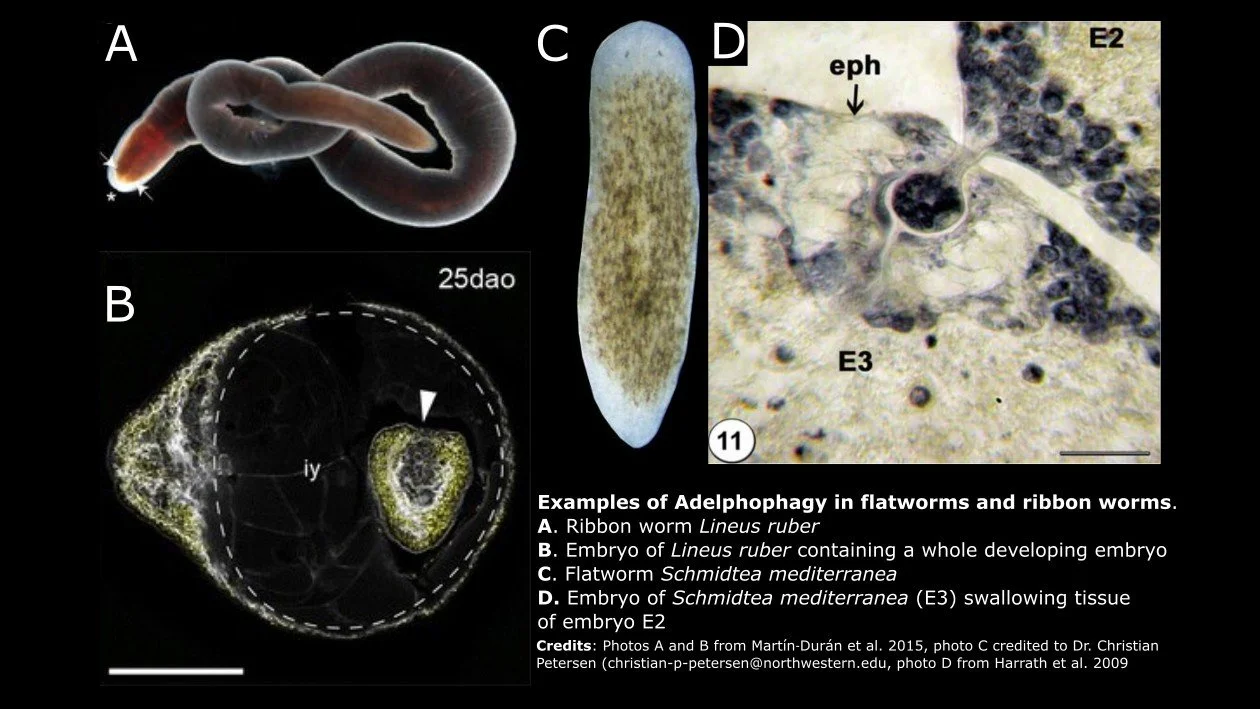
Maternal feeding of siblings also occurs in two types of invertebrates that are unfamiliar to many of us: flatworms and ribbon worms. Both are soft-bodied invertebrates with worm-like bodies; however, they are not true worms. Flatworms belong to the phylum Platyhelminthes, whereas ribbon worms are mostly marine, predatory organisms in the phylum Nemertea.
Martín‐Durán and colleagues (2015) used molecular approaches to demonstrate that the larvae of the ribbon worm Lineus ruber actively feed on both unfertilized eggs and developing siblings. Remarkably, they saw larger individuals that had ingested another developing and apparently viable whole embryo. Ingestion of siblings began before metamorphosis and continued up until post-metamorphic juveniles left their egg string. In 2009, Harrath and colleagues documented adelphophagy for the first time in the planarian Schmidtea mediterranea. In histological slides, they observed some embryos ingesting tissues of adjacent sibling embryos. They could not discern whether entire embryos were consumed or only fragments of tissue. However, many more eggs were deposited in a cocoon than the number of juveniles that eventually hatched, which supports the hypothesis that siblings are consuming whole embryos.
Adelphophagy is remarkable, and a little weird, but it provides valuable insight for comparing early developmental competition and life history strategies across animals. It is rather difficult to study in detail due to the challenges of observing and testing these interactions directly, but further research will be essential to clarify under which ecological or evolutionary factors adelphophagy evolves.
Sources:
Harrath A, Sluys R, Zghal F & Tekaya S. 2009. First report of adelphophagy in flatworms during the embryonic development of the planarian Schmidtea mediterranea (Benazzi, Baguñà, Ballester, Puccinelli & Del Papa, 1975) (Platyhelminthes, Tricladida). Invertebrate Reproduction & Development 53(3), 117–124. https://doi.org/10.1080/07924259.2009.9652297
Martín-Durán JM, Vellutini BC & Hejnol A. 2015. Evolution and development of the adelphophagic, intracapsular Schmidt’s larva of the nemertean Lineus ruber. EvoDevo 6, 28. https://doi.org/10.1186/s13227-015-0023-5
IS THE HEALTH OF THE OFFSPRING REALLY ONLY THE MOTHER’S RESPONSIBILITY?
Decades of research have been dedicated to link the mother’s health and nutritional habits with the health of their children in both the short and the long term. And yes, it is important of course! However, what do we know about the effect of the father’s nutritional habits? Is the father’s only job really to contribute sperm to fertilize the egg? In short, the answers are “yes” and “no”. Yes, because the nutritional habits of the father also affect the offspring. And no, because the sperm does not only contribute genetic material, but also play a fundamental role in fetal and child development.
In an elegant review, Skerrett-Byrne and colleagues (2025) remind us that sperm is far from being only a passive DNA carrier, but rather is an epigenetically active player …
Decades of research have been dedicated to link the mother’s health and nutritional habits with the health of their children in both the short and the long term. And yes, it is important of course! However, what do we know about the effect of the father’s nutritional habits? Is the father’s only job really to contribute sperm to fertilize the egg? In short, the answers are “yes” and “no”. Yes, because the nutritional habits of the father also affect the offspring. And no, because the sperm does not only contribute genetic material, but also play a fundamental role in fetal and child development.
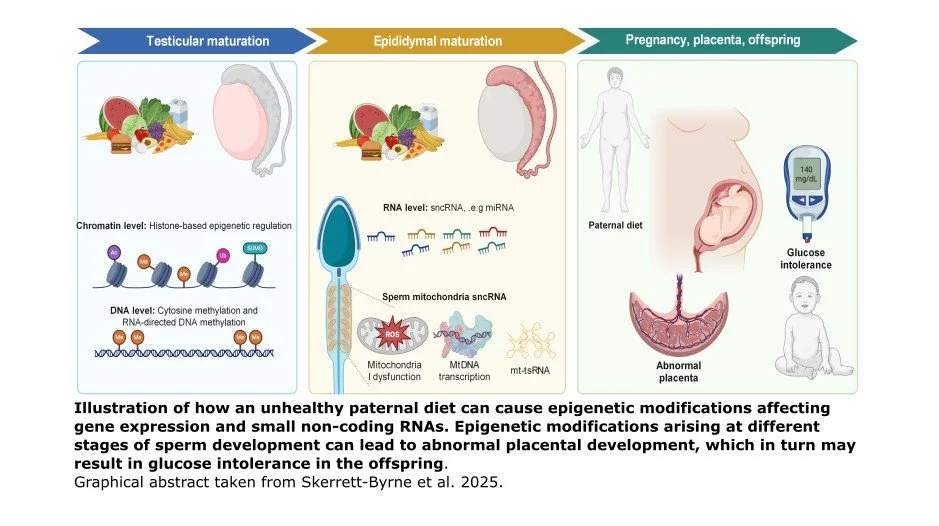
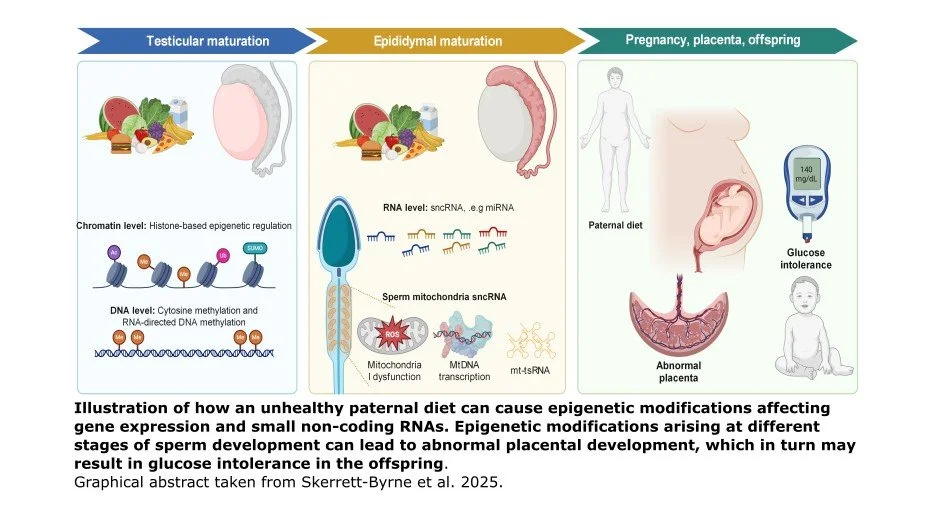
In an elegant review, Skerrett-Byrne and colleagues (2025) remind us that sperm is far from being only a passive DNA carrier, but rather is an epigenetically active player. The epigenome can be thought of as a set of chemical tags that dictate gene expression, turning different genes on and off. In their review, the researchers point out that sperm can acquire epigenetic information, driven by environmental and dietary stressors. They highlight evidence that shows how a high-fat-diet (HFD) causes epigenetic changes in fathers’ sperm that in turn alter the expression of genes and small non-coding RNAs. These changes are implicated in placental and embryonic development, as well as metabolic disease susceptibility in offspring after birth.
The review goes a step further by introducing an interactive digital application, ShinySpermPlacenta, designed to facilitate open-access exploration of the paternal epigenetic data. Users can explore the overlap between HFD sperm epigenetic data and placenta phenotype-driven genes. The app aims to promote systems-level understanding of how preconception environments shape the next generation.
Although the research on the transgenerational effects of paternal nutrition is still new compared to research on maternal nutrition, the growing body of evidence is substantial. Skerrett-Byrne and colleagues highlight the need to reframe preconception care as a truly bi-parental responsibility, rather than placing the entire burden on mothers. This fresh perspective could improve fertility and reduce pregnancy complications and chronic disease in our society.
Source:
Skerrett-Byrne DA, Pepin AS, Laurent K, Beckers J, Schneider R, de Angelis MH & Teperino R. 2025. Dad’s diet shapes the future: How paternal nutrition impacts placental development and childhood metabolic health. Molecular Nutrition & Food Research, 0:e70261. https://doi.org/10.1002/mnfr.70261
TOXIC SEX: WHEN POISON BECOMES A GIFT
Blister beetles, as their name suggests, are beetles of the Meloidae family known for causing blisters upon contact with human skin. Toxicity in these coleopterans is due to the presence of a defensive chemical compound in their hemolymph called cantharidin, which is known to be toxic to mammals, birds, and frogs. Cantharidin has fascinated scientists and human societies alike for centuries and has a long history in both scientific and popular literature.
Female blister beetles contain less cantharidin than males. They are unable to synthesize it themselves, but the way that they acquire it is fascinating: they…
Blister beetles, as their name suggests, are beetles of the Meloidae family known for causing blisters upon contact with human skin. Toxicity in these coleopterans is due to the presence of a defensive chemical compound in their hemolymph called cantharidin, which is known to be toxic to mammals, birds, and frogs. Cantharidin has fascinated scientists and human societies alike for centuries and has a long history in both scientific and popular literature.
Female blister beetles contain less cantharidin than males. They are unable to synthesize it themselves, but the way that they acquire it is fascinating: they receive it from males! Cantharidin is synthesized in the accessory reproductive glands of male blister beetles. The males incorporate it into the spermatophore, and then it is transferred as a nuptial gift during copulation. Females then absorb the cantharidin and incorporate it into their bodies, thereby achieving chemical defense.
Another extraordinary aspect of this process is that females accumulate cantharidin in their ovaries and add it to their eggs. Therefore, blister beetle eggs are also chemically protected against predation. This system is particularly interesting because it is a case in which the same material is socially transferred in two different contexts: from male to female during mating and from female to eggs during maternal provisioning.
In human society, cantharidin has been known for over 2000 years. It has been used in traditional medicine and pharmacology in a multitude of cultures and countries, as an aphrodisiac, a drug, and even a poison. Derivatives of cantharidin are currently being investigated as therapeutic alternatives due to their strong potential as novel anticancer agents. Cantharidin itself was recently approved by the FDA (as a topical solution called “YCANTH”) to treat molluscum contagiosum. The blister beetle-cantharidin system is a great example of how one can derive useful benefits, here in a medical context, from the wonders of nature and the powers of Socially Transferred Materials.
Recommended literature:
McCormick JP, Carrel JE, 1987. Cantharidin biosynthesis and function in meloid beetles. In: Prestwitch, GD, Blomquist, GJ. (Eds.), Pheromone Biochemistry. Academic Press, Orlando, FL, pp. 307–350. https://doi.org/10.1016/B978-0-12-564485-3.50015-4
Muzzi M, Mancini E, Fratini E, Cervelli M , Gasperi T, Mariottini P, Persichini T, Bologna MA & Di Giulio, A. 2022. Male Accessory Glands of Blister Beetles and Cantharidin Release: A Comparative Ultrastructural Analysis. Insects 13, 132. https://doi.org/10.3390/insects13020132
Scott KA, McCluskey A, Njardarson JT & Wang W. 2026.On the history, synthesis, and medicinal use of cantharidin, LB-100, and their analogs. Royal Society of Chemistry Medical Chemistry 17, 743–767. https://doi.org/10.1039/d5md00917k
Sierra JR, Woggon WD, Schmidt H, 1976. Transfer of cantharidin during copulation from the adult male to the female Lytta vesicatoria (Spanish flies). Experientia 32, 142–144. https://doi.org/10.1007/BF01937729
AN OVIPAROUS CAECILIAN FEEDS ON OVIDUCTAL MILK
When you think about amphibians, caecilians are probably not the first animals that come to mind. They are among the most poorly understood amphibians, largely due to their fossorial lifestyle. Classified in the order Gymnophiona, they differ from frogs and salamanders because they are limbless and elongated, resembling snakes or worms more than a “typical” amphibian.
Despite their secretive nature, researchers have documented interesting parental feeding mechanisms, such as…
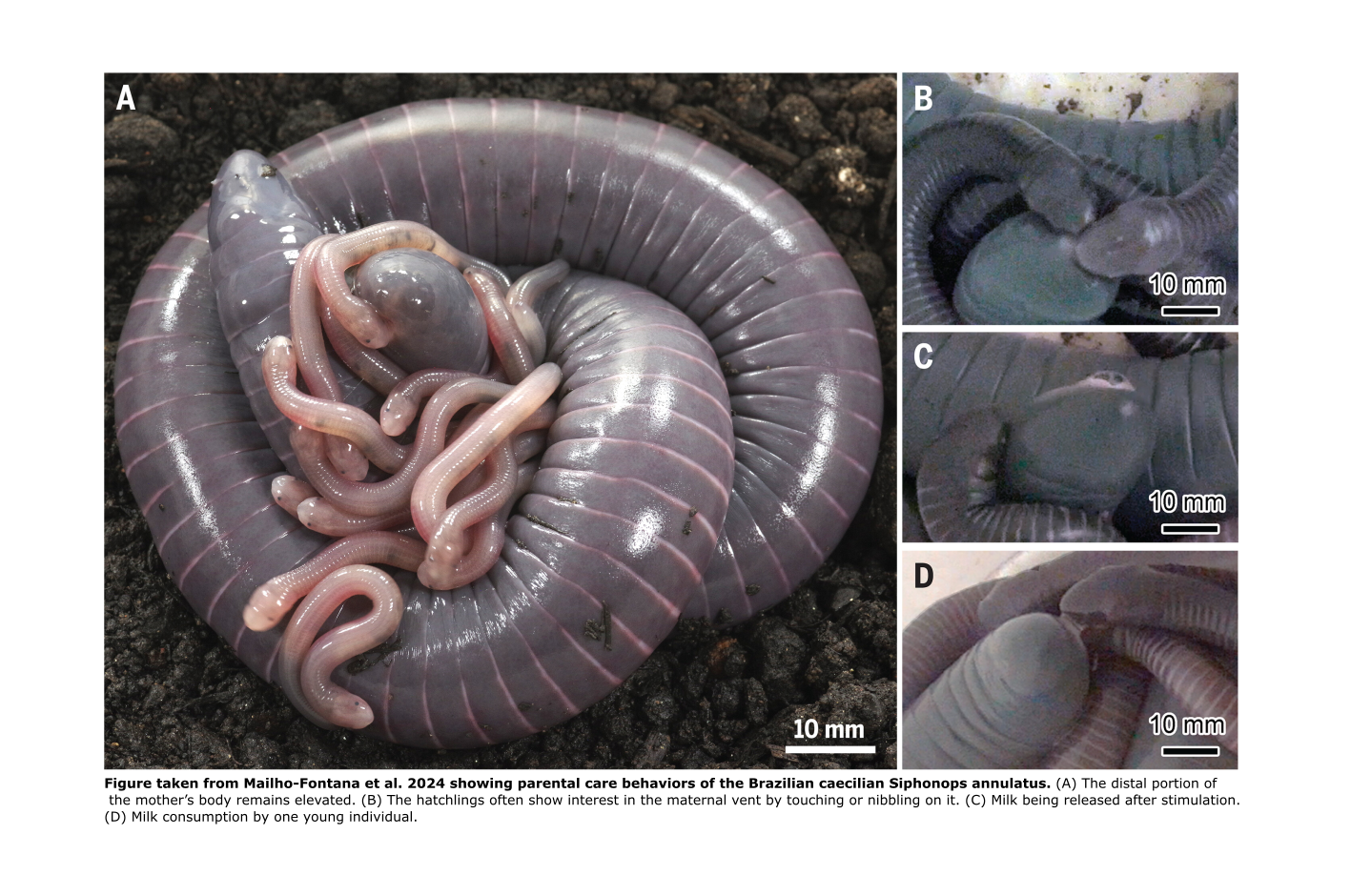
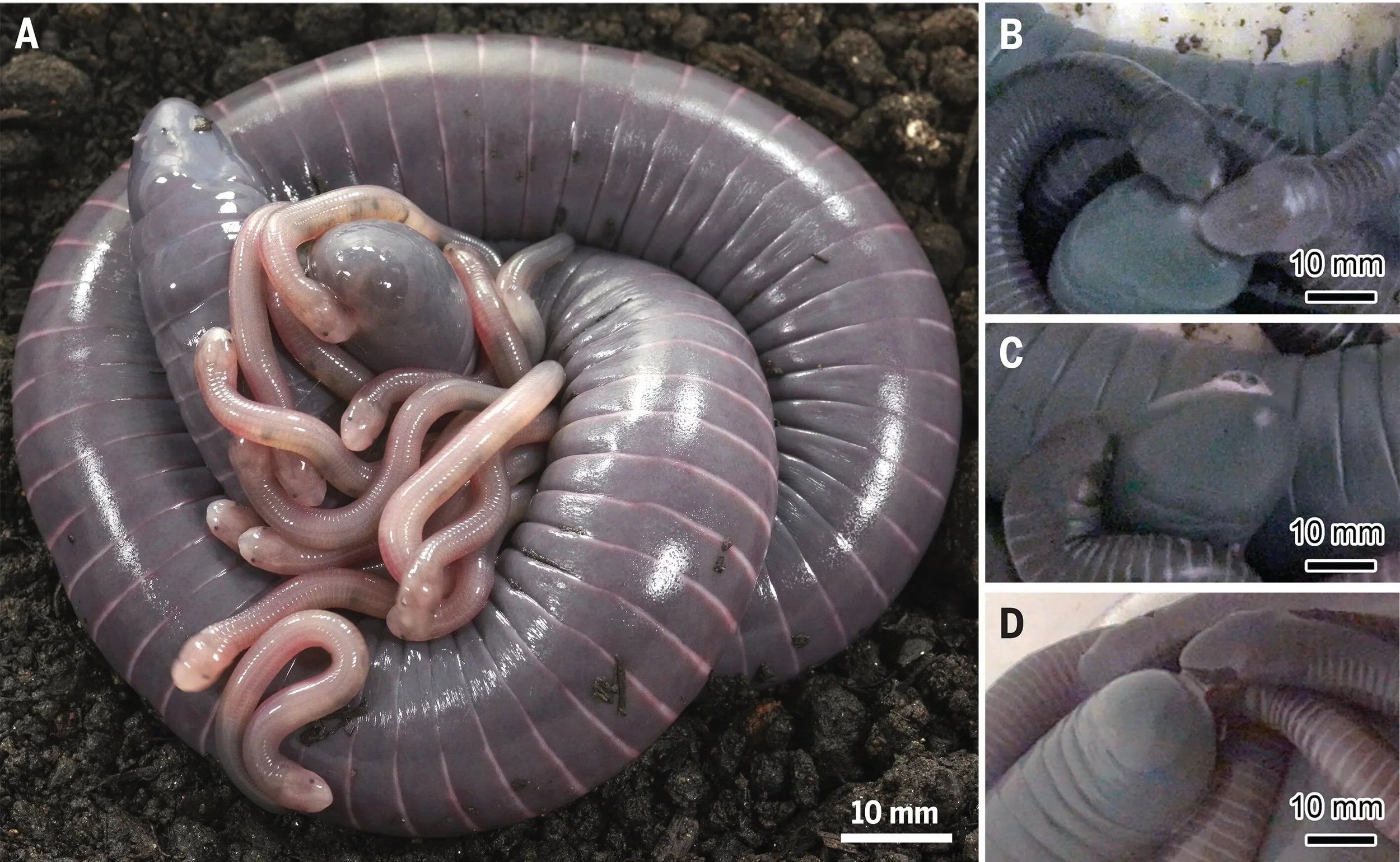
Figure taken from Mailho-Fontana et al. 2024 showing parental care behaviors of the Brazilian caecilian Siphonops annulatus. (A) The distal portion of the mother’s body remains elevated. (B) The hatchlings often show interest in the maternal vent by touching or nibbling on it. (C) Milk being released after stimulation. (D) Milk consumption by one young individual.
When you think about amphibians, caecilians are probably not the first animals that come to mind. They are among the most poorly understood amphibians, largely due to their fossorial lifestyle. Classified in the order Gymnophiona, they differ from frogs and salamanders because they are limbless and elongated, resembling snakes or worms more than a “typical” amphibian. Despite their secretive nature, researchers have documented interesting parental feeding mechanisms, such as nourishment within the oviducts in live-bearing species and skin feeding in oviparous species.
However, this fascinating group keeps surprising us with more forms of parental feeding. Recently, Mailho-Fontana et al. (2024) documented milk provisioning in the Brazilian oviparous caecilian Siphonops annulatus. The mothers exude a secretion from the vent, where the hatchlings gather and feed. It seems like the hatchlings stimulate the female both through physical contact -– touching the vent with their snouts or nibbling – and through high-pitched vocalizations.
Besides milk provisioning, this species also exhibits skin feeding, first described in 2008 by Wilkinson and colleagues. However, this behavior appears to be less frequent, occurring weekly, while milk is provided daily. Milk is clearly important for the nutrition of the young.
But, the researchers did not stop there! They also found that the milk originates from the oviductal lumen and glands that develop from hypertrophied oviductal epithelial cells during the parental care phase. Using histological and chemical analyses, the researchers identified that the milk is composed of neutral glycoconjugates and lipids, a composition similar to the nutritive fluid found in the oviducts of live-bearing caecilians.
Mailho-Fontana et al. (2024) emphasize the importance of integrative studies that join together fieldwork, life history, behavior, and morphology when dealing with animals that have fossorial lifestyles. Something remarkable about their finding is that it is the first time that oviductal milk production has been documented in an oviparous amphibian, which exemplifies the complexity of the evolution of reproductive modes in vertebrates.
The article’s supplementary material (https://doi.org/10.1126/science.adi5379) includes fascinating videos that clearly show the release of milk from the maternal vent and the feeding behavior of the hatchlings.
Source:
Mailho-Fontana PL, Antoniazzi MM, Coelho GR, Pimenta DC, Fernandes LP, Kupfer A ED Brodie Jr, Jared C. (2024). Milk provisioning in oviparous caecilian amphibians. Science 383, 1092–1095. https://doi.org/10.1126/science.adi5379
MILK PROVISIONING IN TSETSE FLIES
You probably have heard about the tsetse fly, the African fly which feeds on blood of both humans and animals. They are a troubling public health concern because they are vectors of African trypanosomes. However, tsetse flies are also known for something else - something extraordinary.
Similar to humans, tsetse flies possess milk glands! Mother flies give birth to live young, after larvae develop inside of their uterus. To nourish their larvae …
You probably have heard about the tsetse fly, the African fly which feeds on blood of both humans and animals. They are a troubling public health concern because they are vectors of African trypanosomes. However, tsetse flies are also known for something else - something extraordinary.
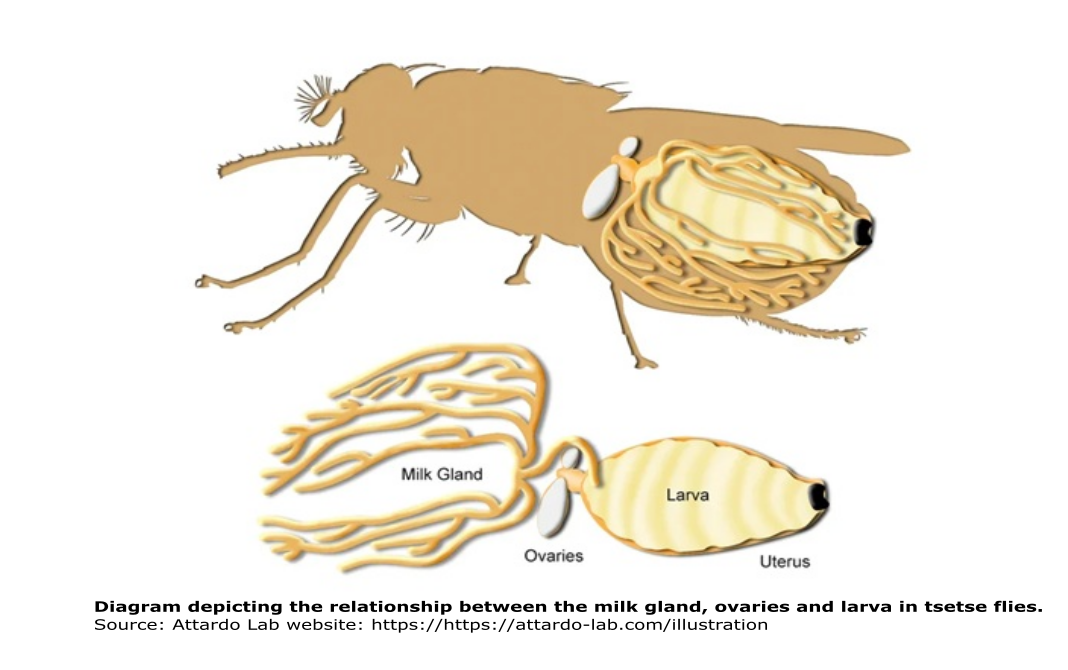
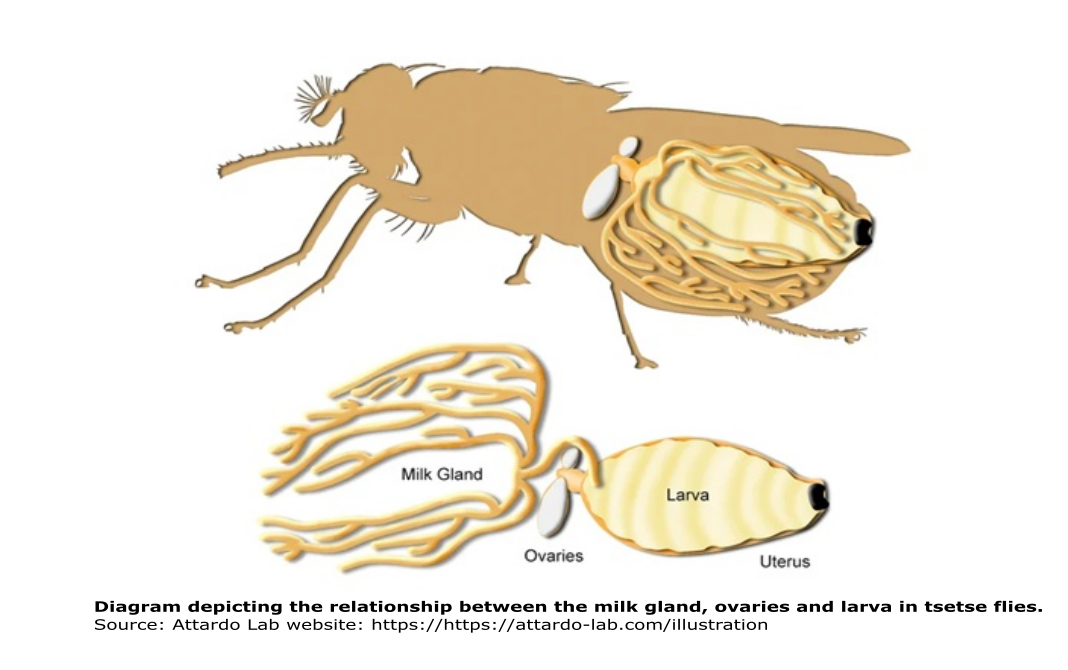
Similar to humans, tsetse flies possess milk glands! Mother flies give birth to live young, after larvae develop inside of their uterus. To nourish their larvae while in the uterus, female tsetse flies use a modified accessory milk gland, which provides proteins and lipids needed for larval development.
In a functional analysis study, Attardo and collaborators (2008) knocked down a milk protein in pregnant female tsetse flies to understand its function in larval nutrition. The knockdown group had a lower rate of pupal deposition relative to the control group in which synthesis of the milk protein was not disrupted. The results suggest that the protein plays an important role in larvigenesis, providing not only nutrition, but also important biochemical function as a carrier of lipid and/or sterol type molecules.
But there is more, a 2-for-1 on socially transferred materials! Along with the milk, mother flies also transmit some bacterial symbionts to their larvae. Balmand and collaborators (2013) found that some symbionts are maternally transferred via the milk glands. These symbionts colonize the lumen of the milk glands and are transferred to the larvae along with the milk. The microbes are important because they might affect the insect’s capacity to act as a vector of trypanosomes. Also, given the incomplete nutritional value of blood, the symbionts are essential in complementing milk to maintain the tsetse fly life cycle.
Watch this incredible video from KQED: https://www.kqed.org/science/1956004/a-tsetse-fly-births-one-enormous-milk-fed-baby
Sources:
Attardo GM, Lohs C, Heddi A, Alam UH, Yildirim S & Aksoy S. (2008). Analysis of milk gland structure and function in Glossina morsitans: Milk protein production, symbiont populations and fecundity. Journal of Insect Physiology 54, 1236–1242. doi.org/10.1016/j.jinsphys.2008.06.008
Balmand S, Lohs C, Aksoy S & Heddi A. (2013). Tissue distribution and transmission routes for the tsetse fly endosymbionts. Journal of Invertebrate Pathology 112, S116–S122. doi.org/10.1016/j.jip.2012.04.002
ORAL SEX IN SPIDERS
When we think about sexual rituals in insects, perhaps the first things that come to mind are sexual cannibalism of males by females, nuptial gifts from males to females, opportunistic mating, pair bonding, genital mutilation, or plugging. All of these, while impressive, are relatively well documented.
But, in 2016, Gregoric and colleagues studied the sexual behavior of the Darwin's bark spider from Madagascar …
When we think about sexual rituals in insects, perhaps the first things that come to mind are sexual cannibalism of males by females, nuptial gifts from males to females, opportunistic mating, pair bonding, genital mutilation, or plugging. All of these, while impressive, are relatively well documented.
But, in 2016, Gregoric and colleagues studied the sexual behavior of the Darwin's bark spider from Madagascar. Among the rituals in the diverse and striking sexual repertoire of this species, researchers documented the occurrence of oral sex. That's right! Males of this species obligatorily secrete oral fluids (saliva) into the female genitalia before, during, and after copulation. How’s that for a socially transferred material!
The exact behavioral and adaptive significance of oral contact in the sexual repertoire of the Darwin's bark spider, is yet to be understood. However, the researchers point to two plausible functions of this behavior. By mechanisms of cryptic choice, females might assess male quality and bias the fertilization of their gametes to favor paternity with sperm from better quality males. Another hypothesis suggests that in order to reduce sperm competition, salivary enzymes might induce some sort of physiological advantage to the donor's sperm over the sperm of rivals. However, It is necessary to decipher the composition of male spider saliva before we can determine the significance of this behavior as well as how often it occurs among spiders, or even other animals.
Here you can watch a video of this behavior, posted by the Evolutionary Zoology Lab: https://www.youtube.com/watch?v=_Zp4-tjha6M. It shows the complete behavior, including the transfer of liquid into the female genitals!!!
Source:
Gregorič M, Šuen K, Cheng RC, Kralj-Fišer S & Kuntner M. (2016). Spider behaviors include oral sexual encounters. Scientific Reports 6, 25128. doi.org/10.1038/srep25128
FEEDING ON SLIME
Velvet worms are among the most extraordinary animals on earth! With a very simple body structure, these ancient animals have been living on our planet for nearly 500 million years. Despite their name, they are not actually worms. They are not insects either, but look like caterpillars, and belong to their own phylum called Onycophora.
Onycophorans have a remarkable technique for hunting. To capture their prey, velvet worms eject a sticky, adhesive slime from their oral papillae, which are located close to their mouth. The sticky slime immobilizes the prey so that the velvet worm can then casually approach it.
In a study of feeding behavior conducted in Costa Rica in 2019, researchers documented for the first time a form of parental feeding in velvet worms...
Velvet worms are among the most extraordinary animals on earth! With a very simple body structure, these ancient animals have been living on our planet for nearly 500 million years. Despite their name, they are not actually worms. They are not insects either, but look like caterpillars, and belong to their own phylum called Onycophora.
Onycophorans have a remarkable technique for hunting. To capture their prey, velvet worms eject a sticky, adhesive slime from their oral papillae, which are located close to their mouth. The sticky slime immobilizes the prey so that the velvet worm can then casually approach it.
In a study of feeding behavior conducted in Costa Rica in 2019, researchers documented for the first time a form of parental feeding in velvet worms involving a socially transferred material. Barquero-Gonzales and colleagues showed that in some species, during the first two weeks after birth, the young only eat one thing: the threads of slime used by their parents to capture their prey! This is an unusual form of parental care, and noteworthy as an example of early evolution of a socially transferred material given how ancient these creatures are.
The slime is composed of proteins and lipids, and although its original function may have been defense and prey capture, the components of the slime also represent a good quality food source. So far this is the only study documenting this behavior in velvet worms, so more work is needed to understand the nutritional importance of slime feeding in young velvet worms.
Source: Barquero-González JP, Vega-Hidalgo A & Monge-Nájera J. (2019). Feeding behavior of Costa Rican velvet worms: food hiding, parental feeding investment and ontogenetic diet shift (Onychophora: Peripatidae). doi.org/10.22458/urj.v11i2.2195